Summary
A method for accurate and non-invasive assessment of the pressure drop in blood flow using time-varying 3D blood velocity data acquired via phase contrast MRI.
What
A technology for estimating pressure drops within the cardiovascular system from 3D blood flow imaging. The estimate can predict pressure differences also in the presence of turbulent blood flow. The technology can be used for improving risk stratification and clinical decision-making in several conditions, including vessel stenosis and hearth conditions, such as valve stenosis or hypertrophic cardiomyopathy.
Why
Pressure drops or differences in relative pressure provide valuable clinical biomarkers for assessing cardiovascular diseases.
To date, catheterisation is still considered the gold standard for measuring pressure drop in the cardiovascular system. However, catheterisation is an invasive technique associated with high risks of infection and high costs.
Other known alternative methods for estimating blood pressure drop, even when it is turbulent, are based on computer simulations (CFD) of the three-dimensional blood flow in vascular structures. However, the simulations require time, a skilled engineer and high computational resources, and their results are dependent on anatomical details and boundary conditions that are difficult to extract from images
Benefits
The proposed technology allows to overcome the problems with the known methods.
Specifically, the technology enables to accurately estimate pressure drop in turbulent and non-turbulent blood flows from 3D, time-varying, blood flow images, such as the ones acquired through PC-MRI. The method can thus be used for accurate prediction of relative pressure drops through complex multibranched blood vessels.
The method can be easily integrated in existing PC-MRI software and/or in advanced PACS software. Its implementation can take less than five seconds to estimate the pressure drop.
The method allows to comprehensively study the pressure along the vessel, circumventing the lack of control of location of catheterised pressure sensors.
Opportunity
The technology has been tested and validated in several body districts under different flow regimes, including turbulent/transitional flow within the ventricle, in patients with dilated cardiomyopathy, in patients at risk of aortic dissection, and in cerebral vessels.
The technology is protected by a European patent application, a Chinese patent application, a US granted patent and two US patent applications, and is available for licensing. Suitable commercial partners are sought for integration and commercialisation of the technology.
The Science
The blood flow within a vascular section is acquired with three-dimensional imaging techniques, such as 4D PC-MRI. The geometry of the vascular section is also acquired and reconstructed via segmentation.
A divergence-free, three-dimensional velocity field of the blood flow is then computed from the acquired data, by imposing the no-slip condition at the walls of the vascular section (i.e. that the velocity of the flow at the walls is null). The computed velocity field is called virtual field.
The pressure drop across any point of the vascular section can thus be estimated as a function of the computed virtual field. This function is called vWERP, and takes into account the kinetic, the advective and the viscous flow behaviour of the virtual field.
In the presence of turbulent flow, a correction factor can be added to the computed expression, which considers also the stochastic flow fluctuations in the fluid flow. Specifically, this correction factor can depend on the covariance of the measured blood flow. Therefore, the function for calculating the pressure drop in turbulent flows (vWERP-t) can be based on the kinetic, the advective and the viscous flow behaviour of the virtual field, plus a virtual turbulent energy dissipation term, which depends on the covariance of the measured blood flow.
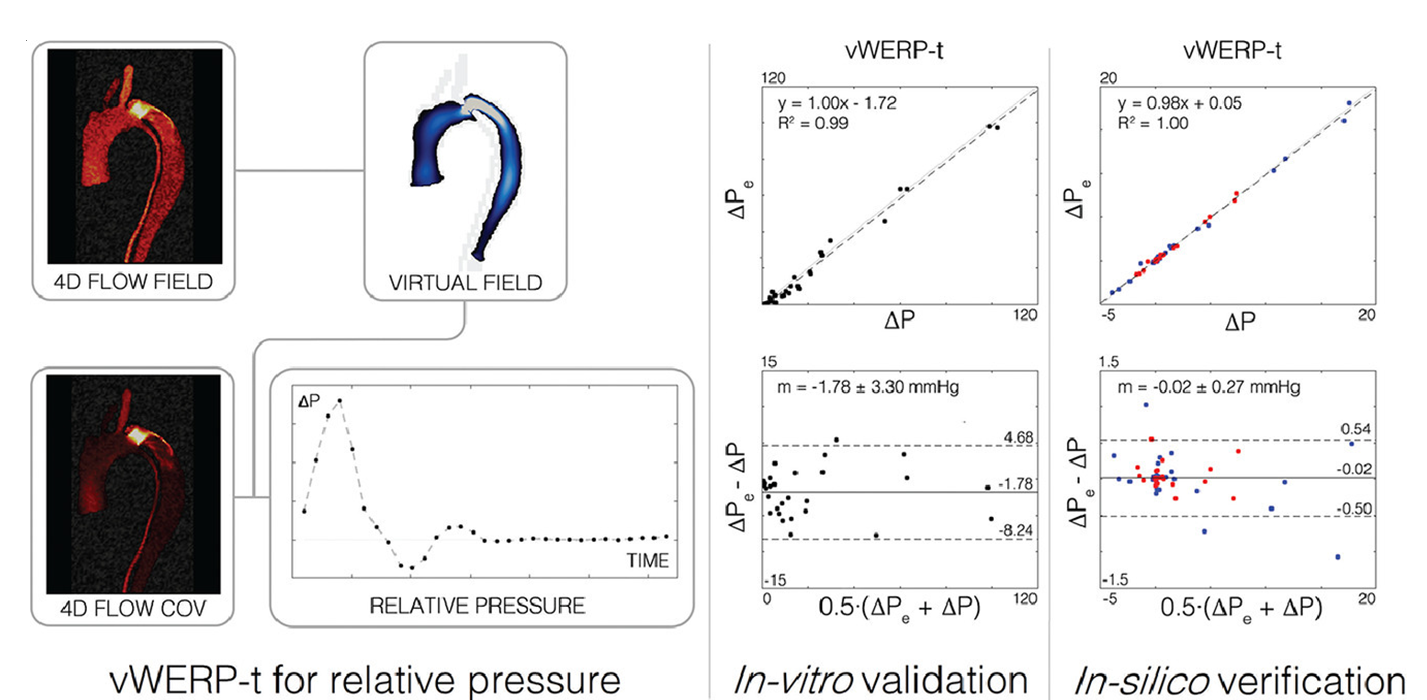
Figure Estimation of the pressure drop (i.e. relative pressure) in an vascular segment with a challenging dissection of the aorta. The pressure estimate has been calculated as a function of the virtual field, considering the correction factor for turbulent blood flows (vWERP-t).
IP Status
The technology is protected by:
Further Information